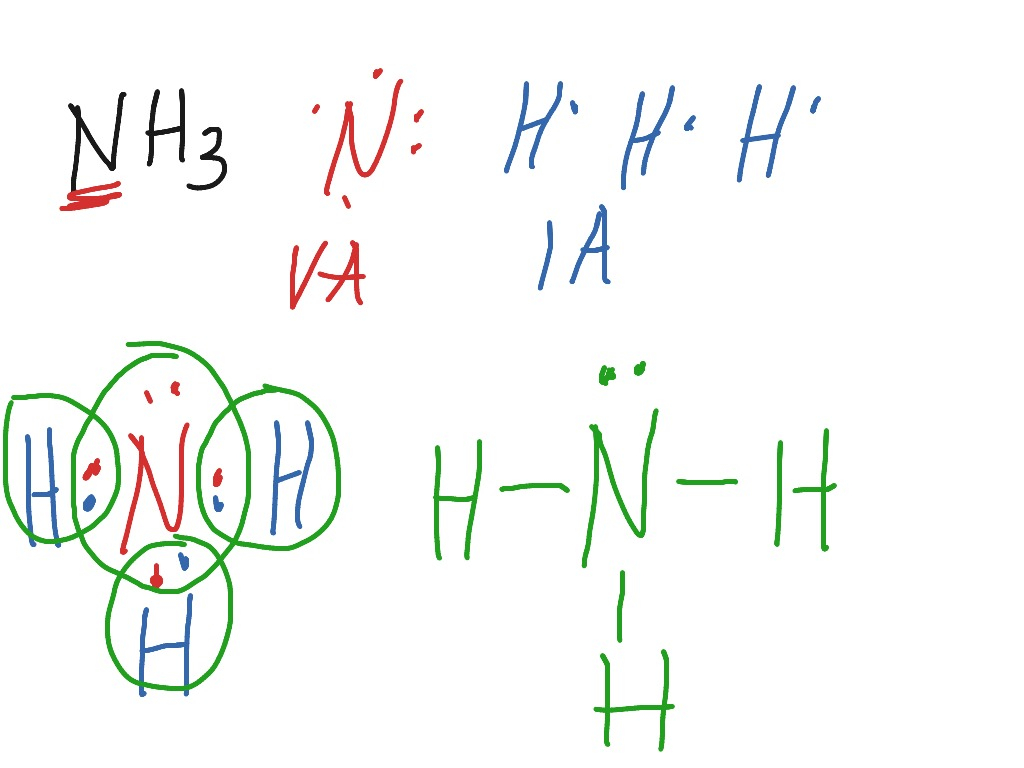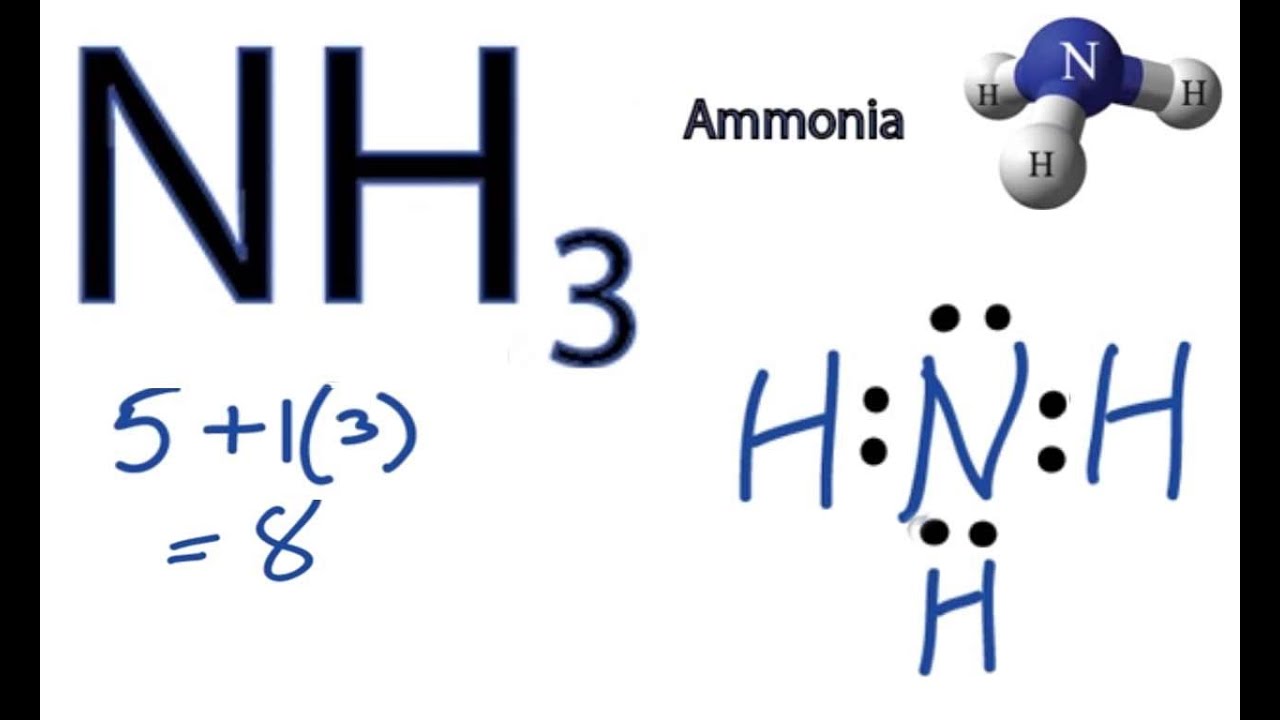Draw The Lewis Structure Of Nh3
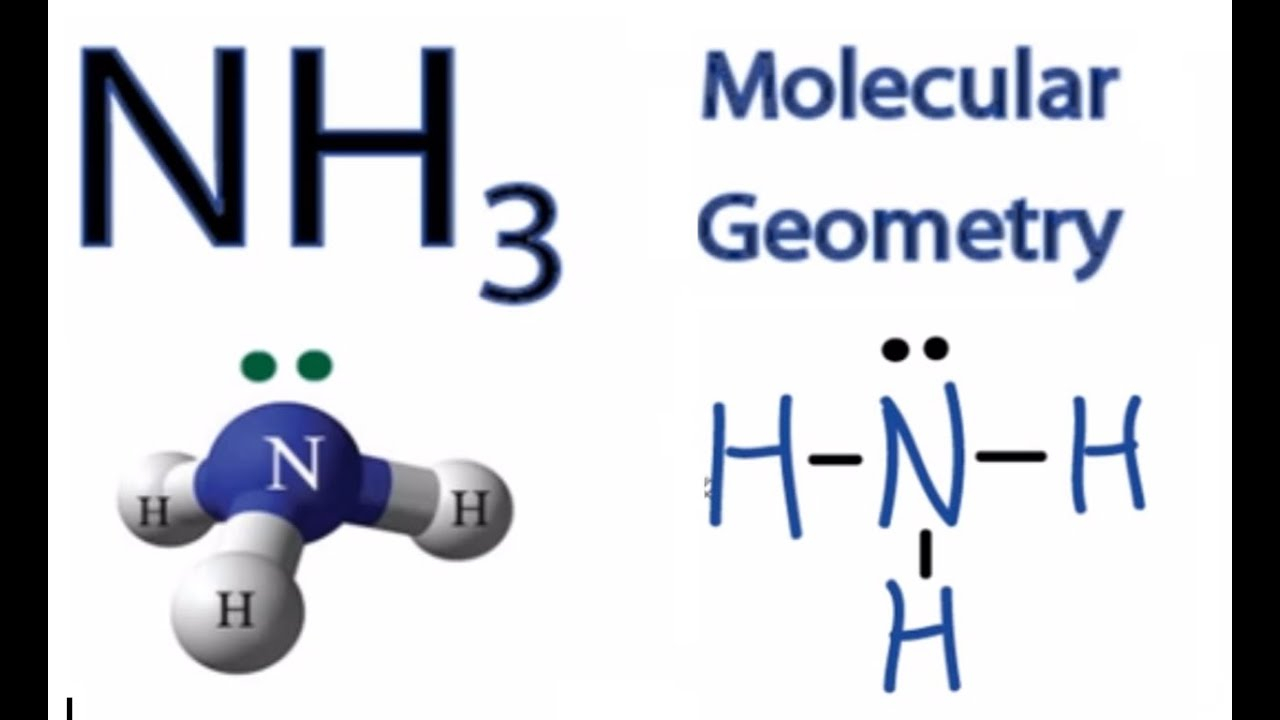
Draw The Lewis Structure Of Nh3 - Web ammonia (nh 3) is a commonly tested lewis structure due to it's widespread use in agriculture as a fertilizer. Now that we know the valence electrons for the molecule, we can predict its lewis. On the periodic table, nitrogen is in group 5 or 15 so it has 5 valence electrons, and then. Nh 3 is made up of two atoms and both have a total of eight electrons in the valance. Lewis structure of nh3 can be drawn by using valence electrons of nitrogen and. Web the lewis structure of ammonia, #nh_3#, would be three hydrogen atoms bonded to a nitrogen atom in the middle, with a lone pair of electrons on top of the atom. Web 6 steps to draw the lewis structure of nh3 step #1: There are 3 single bonds between the. Calculate the total number of valence electrons. Web drawing the nh3 lewis structure involves following a set of rules and guidelines. Web to illustrate this method, let’s calculate the formal charge on the atoms in ammonia (nh3) whose lewis electron structure is as follows: Calculate the total number of valence electrons. Web to draw the nh3 lewis structure (ammonia) involves a few straightforward steps. Now that we know the valence electrons for the molecule, we can predict its lewis. Web a video explanation of how to draw the lewis dot structure for ammonia, along with information about the compound including formal charges, polarity, hybrid orbitals, shape, and bond angles. Web i quickly take you through how to draw the lewis structure of ammonia, nh3. Determine the total number of valence electrons for all the atoms in nh3, which is 5 (for nitrogen) + (3 x 1) (for. Lewis structure of nh3 can be drawn by using valence electrons of nitrogen and. Web we're going to do the lewis structure for nh3: This will help you understand the molecule’s electronic structure and bonding. Nh 3 is made up of two atoms and both have a total of eight electrons in the valance. Web to draw the nh3 lewis structure (ammonia) involves a few straightforward steps. Web steps to draw the lewis structure of nh 3. Web to draw the nh3 lewis structure, follow these steps: Web to illustrate this method, let’s calculate the. Calculate the total number of electrons. There are 3 single bonds between the. On the periodic table, nitrogen is in group 5 or 15 so it has 5 valence electrons, and then. It also is a good example of a molecule with a trigonal prymidal. Web understanding the nh3 lewis structure is crucial for comprehending the chemical properties and behavior. Web to draw the nh3 lewis structure, follow these steps: Web we're going to do the lewis structure for nh3: The following procedure will give you the correct lewis structure for any. Web i quickly take you through how to draw the lewis structure of ammonia, nh3. Now that we know the valence electrons for the molecule, we can predict. There are 3 single bonds between the. Web in this video i draw the dot and cross diagram for nh3 (ammonia). Web in today's video, i draw out the lewis dot structure for nh3, commonly known as ammonia. Web 6 steps to draw the lewis structure of nh3 step #1: Web the lewis structure of ammonia, #nh_3#, would be three. I also go over hybridization and bond angle. Web a video explanation of how to draw the lewis dot structure for ammonia, along with information about the compound including formal charges, polarity, hybrid orbitals, shape, and bond angles. Web drawing the nh3 lewis structure involves following a set of rules and guidelines. A neutral nitrogen atom has five valence. Web. Web to draw the nh3 lewis structure, follow these steps: Determine the total number of valence electrons for all the atoms in nh3, which is 5 (for nitrogen) + (3 x 1) (for. Web ammonia (nh 3) is a commonly tested lewis structure due to it's widespread use in agriculture as a fertilizer. Web to illustrate this method, let’s calculate. Web ammonia (nh 3) is a commonly tested lewis structure due to it's widespread use in agriculture as a fertilizer. Web to draw the nh3 lewis structure (ammonia) involves a few straightforward steps. There are 3 single bonds between the. I also go over hybridization and bond angle. Web understanding the nh3 lewis structure is crucial for comprehending the chemical. Web to draw the nh3 lewis structure (ammonia) involves a few straightforward steps. Now that we know the valence electrons for the molecule, we can predict its lewis. Web drawing the nh3 lewis structure involves following a set of rules and guidelines. Lewis structure of nh3 can be drawn by using valence electrons of nitrogen and. This is a clip. It also is a good example of a molecule with a trigonal prymidal. Web understanding the nh3 lewis structure is crucial for comprehending the chemical properties and behavior of ammonia. This will help you understand the molecule’s electronic structure and bonding. Web we're going to do the lewis structure for nh3: Nh 3 is made up of two atoms and. Web in today's video, i draw out the lewis dot structure for nh3, commonly known as ammonia. Web in this video i draw the dot and cross diagram for nh3 (ammonia). Web 6 steps to draw the lewis structure of nh3 step #1: Calculate the total number of electrons. Nh 3 is made up of two atoms and both have. Web ammonia (nh 3) is a commonly tested lewis structure due to it's widespread use in agriculture as a fertilizer. Web in this video i draw the dot and cross diagram for nh3 (ammonia). Web to draw the nh3 lewis structure, follow these steps: This will help you understand the molecule’s electronic structure and bonding. It also is a good example of a molecule with a trigonal prymidal. Web to draw the nh3 lewis structure (ammonia) involves a few straightforward steps. Web 6 steps to draw the lewis structure of nh3 step #1: This is a clip from the complete video: Web understanding the nh3 lewis structure is crucial for comprehending the chemical properties and behavior of ammonia. Web drawing the nh3 lewis structure involves following a set of rules and guidelines. Nh 3 is made up of two atoms and both have a total of eight electrons in the valance. Web the lewis structure of ammonia, #nh_3#, would be three hydrogen atoms bonded to a nitrogen atom in the middle, with a lone pair of electrons on top of the atom. On the periodic table, nitrogen is in group 5 or 15 so it has 5 valence electrons, and then. There are 3 single bonds between the. Determine the total number of valence electrons for all the atoms in nh3, which is 5 (for nitrogen) + (3 x 1) (for. Web a video explanation of how to draw the lewis dot structure for ammonia, along with information about the compound including formal charges, polarity, hybrid orbitals, shape, and bond angles.Nh3 Lewis Structure Electrons Drawing Easy
The Nh3 Lewis Dot Structure Understanding The Basics vrogue.co
NH3 Molecular Geometry Science Education and Tutorials
ShowMe Lewis Structure for NH3
[Solved] Find the following of the compound NH3 Chemical formula Dot
Lewis Dot Diagram For Nh3 exatin.info
Nh3 Lewis Structure Molecular Geometry
Lewis Dot Diagram For Nh3 exatin.info
NH3 Lewis Structure How to Draw the Dot Structure for NH3 (Ammonia
Draw The Lewis Structure For Nh3 Drawing.rjuuc.edu.np
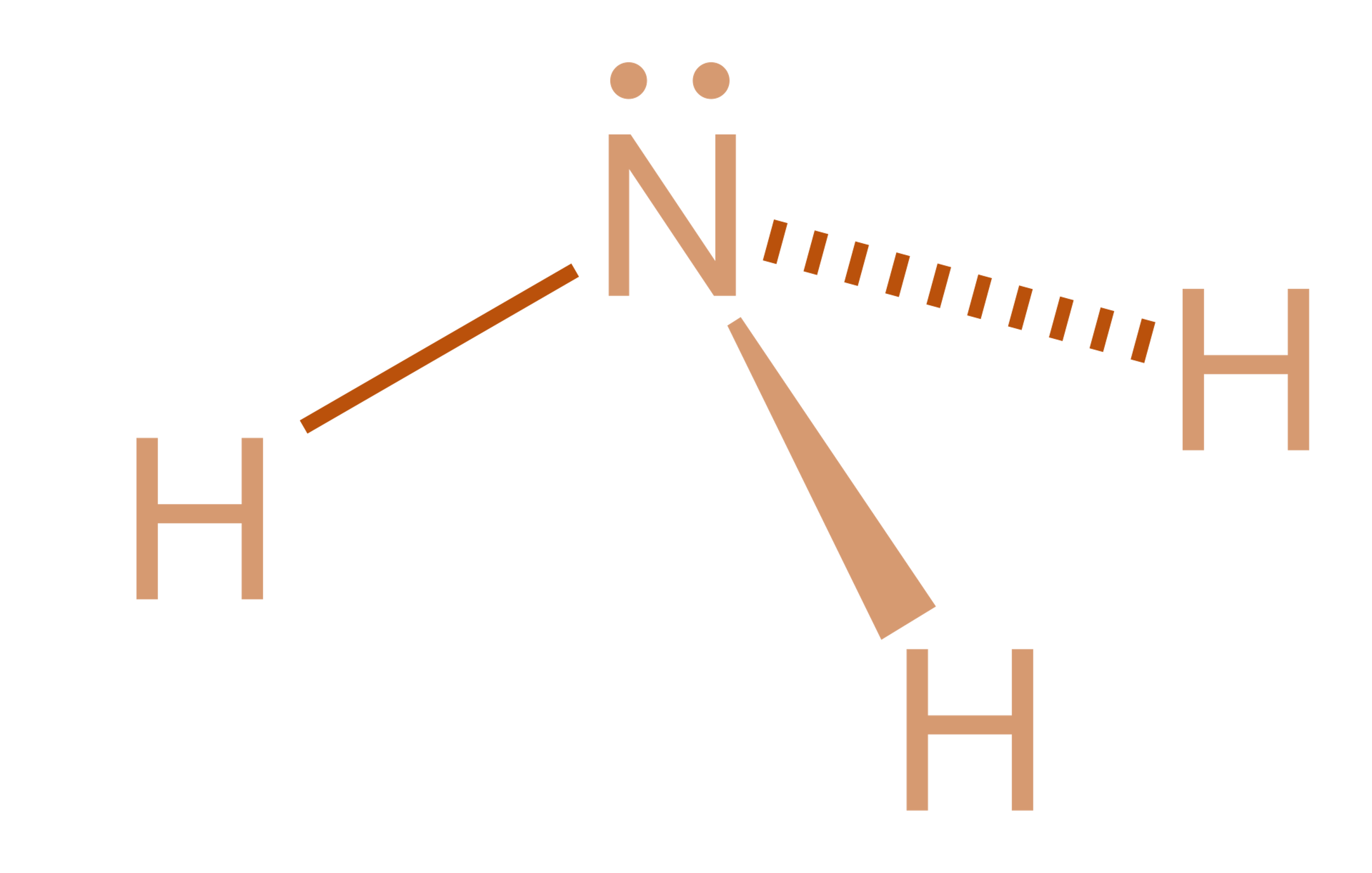
Here, The Given Molecule Is Nh3 (Ammonia).
Now That We Know The Valence Electrons For The Molecule, We Can Predict Its Lewis.
Web We're Going To Do The Lewis Structure For Nh3:
I Also Go Over Hybridization And Bond Angle.
Related Post: